Article
Uncover the hidden benefits of rental-with-equity programs and learn how to turn your lab’s dreams into reality.

This article describes how the Frontier Micro-Furnace Tandem µ-Reactor enables the rapid catalyst screening of laboratory-prepared catalysts, specifically carbon fiber reinforced plastic.
The Micro-furnace Tandem μ-Reactor rapidly screens laboratory-prepared catalysts. A Tandem µ-Reactor consists of a pyrolysis micro-furnace and a catalytic reactor directly connected to a gas chromatograph-mass spectrometer (GC/MS) to observe real-time changes in the gas-phase catalytic products reaction between the sample and the catalyst.
The two reactors (upper and lower) are individually temperature-controlled. The upper reactor (1st Reactor) is used to preheat a gas, vaporize a liquid, or thermally decompose organic solids to generate gases. The catalyst reaction tube is packed with a catalyst and placed in the lower reactor (2nd Reactor).
Volatiles released from the heated or thermally decomposed sample stream flow into the catalyst reaction tube in the lower reactor via carrier gas flow, where they react with the catalyst. Then the products formed flow into the GC for analysis. The catalyst is evaluated by noting what compounds are found and their relative distribution using MS detection. The figure below illustrates a rapid catalyst evaluation system using the MS as the detector.
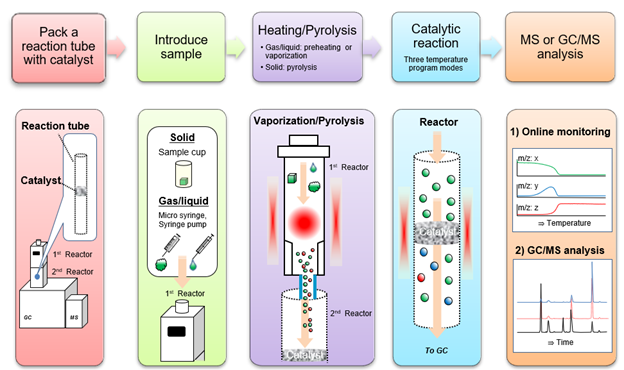
The Tandem µ-Reactors can be used in three different modes of operations. These modes differ in how the reactor temperature is controlled. The isothermal mode has a “Post Heat” function to thermally desorb reaction products in the micro-pores of various types of catalysts. In the other two modes, the furnace temperature can be programmed in either a linear or stepwise progression, up to a maximum of 8 steps.
A short technical note about pyrolysis and reforming of gases by zeolite catalysts of carbon fiber reinforced plastic (CFRP) is described here to demonstrates the capability of this technology.
With the optimum catalyst for the decomposition of epoxy resin, the formation of valuable products is selectively controlled in the reforming of the gases generated by the pyrolysis of CFRP. Previous studies clearly show that BEA catalysts promote the decomposition of bisphenols and suppress the formation of naphthalene.
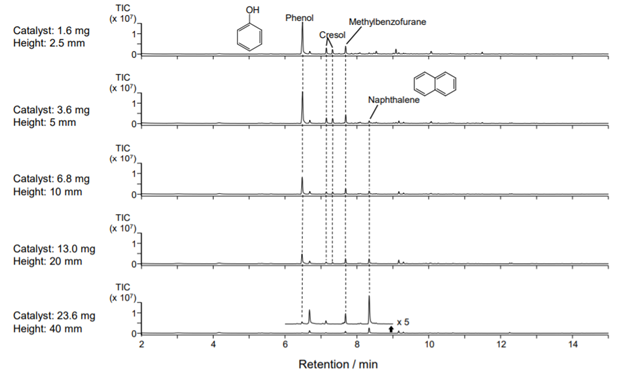
In this note, the optimum reaction conditions for the recovery of phenol which has a higher economic value than the other reaction products, are examined using the BEA catalyst (Si/Al = 92.5). Prepreg (T700SC/2592) was used as the CFRP. 0.3 mg of CFRP was placed in an inert sample cup and introduced into the 1 st Reactor, which was heated at 500 °C. The gases generated flowed directly to a quartz reaction tube (containing the catalyst) in the 2nd Reactor. The catalytic products were separated and detected by GC/MS.
The chromatograms of the products obtained from the catalytic reforming of the gases generated from the pyrolysis of CFRP are shown below. The relative contact time with the catalyst is assumed to be directly related to the height of the packed bed in the reaction tube – assuming a constant flow at each height.
The peak intensity of phenol decreases, and the amount of naphthalene, which is considered to be a secondary product, increases as a function of contact time; thus, mg quantities of the proper catalyst will suppress the formation of naphthalene and increase the yield of phenol [Frontier Laboratories, Ltd.].
References: This technical content was developed by Frontier Laboratories Ltd. 4-16-20 Saikon, Koriyama, Fukushima, 963-8862 JAPAN.
Put our expertise, strategic partnerships, and technical support capabilities to work for you.
8301 New Trails Drive, Suite 100, The Woodlands, Texas 77381
© 2024 Quantum Analytics | A Black Forest Ventures Company | All Rights Reserved | Privacy Policy
Complete this form below to sign up and we will reach out to you with instructions
